|
“Download Wall Painting Paint A Wall Free Photo” (CC0) via Needpix. “Silica gel” By KENPEI – KENPEI' s photo (CC BY-SA 3.0) via Commons WikimediaĢ. “Emulsion Definition and Examples.” ThoughtCo, Feb. they are made out of a specific framework. Dry wood is a classic example of solid foam. If stirred it will mix for a while but soon it will be separated. A golden example of an emulsion is water and oil. However, sometimes we can find some emulsions in the semisolid state according to its application. The emulsion is a mixture of more than one liquid or moire that cannot be mixed. The key difference between gel and emulsion is that a gel is a semisolid substance, whereas an emulsion is a liquid. are examples of emulsions.īelow infographic summarizes the differences between gel and emulsion.Ī gel and an emulsion are two different chemical substances. are some examples of gels while paint, butter, egg yolk, etc. Fruit jelly, gelatin mixture, ointments, etc. However, we can find some emulsions in the semisolid state according to its application. Gel and emulsion are two different chemical substances. What is the Difference Between Gel and Emulsion? We often tend to use the two terms emulsion and colloid interchangeably, but the term emulsion specifically explains the mixture of two liquids that form a colloid. We can describe an emulsion as a mixture of two liquids which are immiscible with each other. What is Emulsion?Īn emulsion is a fine dispersion of minute droplets of one liquid in another in which it is not soluble or miscible. After the completion of the formation of this infinite network material, we can call it a “gel”, and this gel does not dissolve in the solvent. We can detect the gel point of a system easily via observing a sudden change in viscosity. At this point, the mixture loses its fluidity and viscosity and becomes very large. Gelation is a type of crosslinking, and it leads to the formation of a large polymer network.ĭuring the gelation process, a single macroscopic molecule forms at some point, and we call this point as the gel point. In this process, branched polymers cause the formation of linkages between branches. Or how about one of our huge collection of easy chemistry experiments.Gelation is the formation of a gel from a mixture of polymers. You could even give these as a homemade gift afterwards! It is this effect that makes a blue iris look blue, not pigment! Questions to ask about colloids This is because blue visible light ( which has the shortest wavelength ) is scattered by the particles, while longer wavelengths of light are not scattered. The Tyndall Effect also makes clear substances containing tiny particles to appear slightly blue. When a light is shone through a colloidal dispersion the light beam becomes visible as a column of light. The Tyndall effect happens when light is scattered by particles of in its path to create a beam of light.Ĭolloids exhibit the Tyndall effect. This is random zig zag motion of particles that can be seen under a microscope and happens when particles collide into each other in the dispersing medium ( for example – air, water ) Tyndall Effect 
Colloids and Brownian MotionĬolloids also exhibit Brownian movement. Solid -Gas Colloidĭust is made up of solid particles dispersed in the air. If you make jelly with gelatin, this is a colloid of gelatin dispersed in water or juice. Whisk two egg yolks with a little olive oil until you get mayonnaise. This is whipped cream and is a gas-liquid colloid as gas has collected in the liquid forming a stable mixture. This is a special type of colloid known as an emulsion ( milk and cream are also emulsions )Īn emulsion is a special type of colloid made up from a mixture of two liquids which form a stable substance that has different physical properties to the two individual liquids.Ĭolloids can be made up of gas-liquid, liquid-liquid, solid-liquid or solid-gas.Ĭan you think of any more colloid examples for us? Gas – liquid colloid Mayonnaise is a mixture of oil and water, stabilised by proteins in the egg yolk. Butter is also a colloid as there are water molecules trapped in between the fat. If you put cream in a jar and shake for a about 10 minutes the fat molecules stick together, making butter and a liquid called buttermilk. The particles making up a colloid are smaller than those in a suspension. Cream is a colloid as it’s made up of tiny particles of fat dispersed in water. 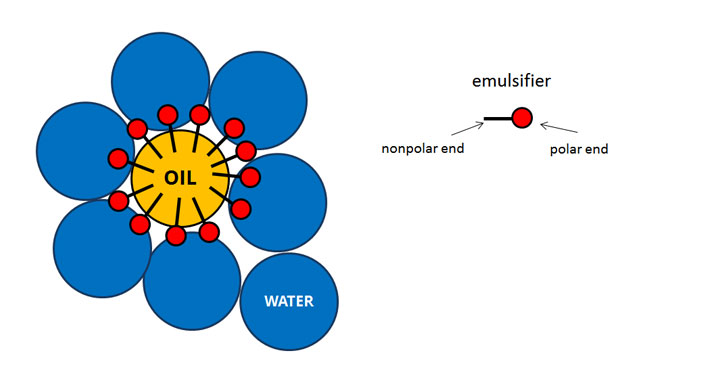
They are a special type of mixture where tiny particles of one substance are scattered through another substance. What is a colloid ? A colloid is a mixture is two or more substances mixed together but not chemically combined ( they can be separated ).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
